
2023 Turf Insect Tracking Data Available Now
In 2022 and 2023, we maintained turf insect trapping sites to monitor adult and caterpillar activity, narrowing our application …


El inglés es el idioma de control de esta página. En la medida en que haya algún conflicto entre la traducción al inglés y la traducción, el inglés prevalece.
Al hacer clic en el enlace de traducción se activa un servicio de traducción gratuito para convertir la página al español. Al igual que con cualquier traducción por Internet, la conversión no es sensible al contexto y puede que no traduzca el texto en su significado original. NC State Extension no garantiza la exactitud del texto traducido. Por favor, tenga en cuenta que algunas aplicaciones y/o servicios pueden no funcionar como se espera cuando se traducen.
Inglês é o idioma de controle desta página. Na medida que haja algum conflito entre o texto original em Inglês e a tradução, o Inglês prevalece.
Ao clicar no link de tradução, um serviço gratuito de tradução será ativado para converter a página para o Português. Como em qualquer tradução pela internet, a conversão não é sensivel ao contexto e pode não ocorrer a tradução para o significado orginal. O serviço de Extensão da Carolina do Norte (NC State Extension) não garante a exatidão do texto traduzido. Por favor, observe que algumas funções ou serviços podem não funcionar como esperado após a tradução.
English is the controlling language of this page. To the extent there is any conflict between the English text and the translation, English controls.
Clicking on the translation link activates a free translation service to convert the page to Spanish. As with any Internet translation, the conversion is not context-sensitive and may not translate the text to its original meaning. NC State Extension does not guarantee the accuracy of the translated text. Please note that some applications and/or services may not function as expected when translated.
Collapse ▲
In 2022 and 2023, we maintained turf insect trapping sites to monitor adult and caterpillar activity, narrowing our application …
View the new Spring 2024 pest update video, covering weed, disease and insect management in …

Grady Miller and Miguel Castillo Ever thought about what would be the ideal grass to use for Donkey pastures? Back …

The turfgrass entomology group has published “Common Insects of Wildflower Plantings within North Carolina Turfgrass: A Field Guide“. This guide …
The updated 2024 manual (2024 Pest Control for Professional Turfgrass Managers) is now available (to view or print PDF) …

Overall, total turfgrass samples submitted in 2023 to the NC State Turf Diagnostics Lab were 11% higher than samples …
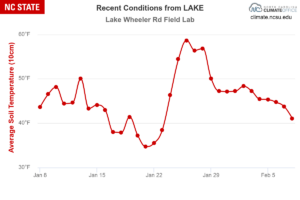
When to Apply Preemergent Herbicides for Turfgrass in North Carolina It is well documented that our winter temperatures are rising …

Regional Turfgrass Conference for Caldwell County is scheduled for April 10 to begin at 8:00 a.m. in Lenoir’s Civic …

Regional Turfgrass Morning Conference for New Hanover County is scheduled for February 28 to begin at 8:00 a.m. in Wilmington. …

Regional Turfgrass Afternoon Conference for New Hanover County is scheduled for February 28 to begin at 1:00 p.m. in …

Regional Turfgrass Conference for Guilford County is scheduled for March 12th, 2024 to begin at 8 a.m. in Greensboro. There …

Regional Turfgrass Conference for Johnston/Wake County is scheduled for March 20th, 2024 to begin at 8 a.m. in Smithfield. There …

***2024 Short Course is FULL. All seats have been taken*** Register now for the 2024 Turfgrass Short Course. The Turfgrass …

The U.S. Environmental Protection Agency (EPA) released its Proposed Interim Registration Review Decision for chlorothalonil (Registration Review Case #0097) …

Purple haze all on my bermudagrass putting greens. Lately, this turf just don’t seem the same. Actin’ funny like …
View the new Winter 2023 pest update video on the TurfFiles website, covering weed, disease and insect management in …

Fred Yelverton, NC State Turfgrass Weed Scientist, received the Distinguished Service Award from the Carolinas Golf Course Superintendents Association …

Don’t miss our fall event, The 4th Annual Turfgrass Research Symposium, where you will hear all things new in …
View the new Fall 2023 pest update video on the TurfFiles website, covering weed, disease and insect management in …

As the summer is coming to a close, except for 100oF temperatures in September, we are in the window …