Center Update: Nitrogen Emissions From Turfgrass
go.ncsu.edu/readext?759777
en Español / em Português
El inglés es el idioma de control de esta página. En la medida en que haya algún conflicto entre la traducción al inglés y la traducción, el inglés prevalece.
Al hacer clic en el enlace de traducción se activa un servicio de traducción gratuito para convertir la página al español. Al igual que con cualquier traducción por Internet, la conversión no es sensible al contexto y puede que no traduzca el texto en su significado original. NC State Extension no garantiza la exactitud del texto traducido. Por favor, tenga en cuenta que algunas aplicaciones y/o servicios pueden no funcionar como se espera cuando se traducen.
Português
Inglês é o idioma de controle desta página. Na medida que haja algum conflito entre o texto original em Inglês e a tradução, o Inglês prevalece.
Ao clicar no link de tradução, um serviço gratuito de tradução será ativado para converter a página para o Português. Como em qualquer tradução pela internet, a conversão não é sensivel ao contexto e pode não ocorrer a tradução para o significado orginal. O serviço de Extensão da Carolina do Norte (NC State Extension) não garante a exatidão do texto traduzido. Por favor, observe que algumas funções ou serviços podem não funcionar como esperado após a tradução.
English
English is the controlling language of this page. To the extent there is any conflict between the English text and the translation, English controls.
Clicking on the translation link activates a free translation service to convert the page to Spanish. As with any Internet translation, the conversion is not context-sensitive and may not translate the text to its original meaning. NC State Extension does not guarantee the accuracy of the translated text. Please note that some applications and/or services may not function as expected when translated.
Collapse ▲Characterization Of Reactive Nitrogen Compounds (Ammonia, Oxides Of Nitrogen, And Nitrous Oxide) From Turfgrass: Their Emissions And Emission Factors Development
PIs: Viney P. Aneja and John T. Walker; Student: Alberth Nahas
Introduction
There are currently no studies in which reactive nitrogen (NH3, NO, and N2O) emissions from turf grass are fully speciated to concurrently examine the individual and total fluxes of NH3, NO, and N2O. Their emissions are rather presented as total nitrogen emitted from the turfgrass system, or measurements are limited to N2O emissions only. The objectives for the project are to i) quantify (both measurements and modeling) emissions of reactive nitrogen compounds (Nr, i.e., ammonia (NH3), nitric oxide (NO), and nitrous oxide (N2O)) from turfgrass systems; ii) quantify relationships between fluxes and turfgrass management activities, including fertilizer type and amount; iii)Validate and utilize process-based EPIC biogeochemical model to elucidate production mechanisms to target mitigation options; and iv) Develop emission factors for Nr from turfgrass and assessment for implications for current inventories.
Methods
The sampling intensives were conducted during the four predominant seasons (~2 weeks each) at the Lake Wheeler Turfgrass Field Laboratory utilizing a dynamic chamber system interfaced to an environmentally controlled mobile laboratory. The sampling site was a 50 ft by 50 ft tall fescue field, divided into 30 individual plots. Each individual plot covers 2.25 m2 with a 60 cm unfertilized margin between each plot and ~3 m wide alley in the middle of the site to accommodate movement of the mobile laboratory. Within each plot, a permanent soil PVC collar was randomly inserted into the soil to a depth of ~15 cm.
The air conditioned mobile lab (modified Ford Aerostar van) was parked at the sampling site to house a chemiluminescence-based Thermo Environmental Instruments Model 17C NH3, NO, and NO2 Analyzer, a Thermo Environmental Instruments Model 146 Dynamic Calibrator, a vacuum pump, a datalogger (CR800 series, Scientific Campbell), two compressed gas calibration standards (NO and NH3, both balanced in N2, Airgas, Raleigh), and two zero-grade air cylinders (Airgas, Raleigh). During the measurement, the dynamic chamber (FEP Teflon-lined, open-bottom white PVC cylinder) was placed on the turfgrass plot and was attached to an air-tight sealed soil collar. Zero air was continuously supplied into the chamber at a constant rate of 1.2 lpm to flush the ambient air from the chamber and to ensure the instrument only analyzed Nr emissions from turfgrass. The chamber was in close proximity to the NH3 converter to minimize NH3 loss from the measurement. The air volume inside the chamber was kept constant (~3900 cm3) to maintain the consistency of measurement. The air residence time inside the chamber is ~3.25 minutes. Also, the turfgrass inside the chamber was clipped so that its height was at the same level as the soil collar. The clipped turfgrass was removed from the chamber to avoid unintended additional Nr emissions to the chamber.
The NO and NH3 concentrations inside the chamber were measured in near real time. The concentration data analyzed by the instrument were stored in the datalogger. Concentrations from each plot were measured for approximately 68 minutes. The first eight minutes of the measurement were intended to equilibrate the air inside the chamber, ensuring the ambient air was flushed out by zero air. Thus, the data for this period are not considered and an interval of one hour was used for further analysis. Meanwhile, 20 ml of the chamber outlet air was collected in glass vials (Labco, UK) for N2O analysis in the laboratory at the U.S. EPA (Research Triangle Park, NC). The N2O concentrations were measured by using gas chromatography equipped with electron capture detecting method. The air samples for N2O concentrations (and subsequent emission analysis) were collected at 20, 40, and 60 minutes during the 1-hour chamber measurement interval.
Results and Discussions
Ammonia emissions are expected to be the highest among the three species. This is because the fertilizer used in this study is NH3-based. Overall, NH3 emissions accounts for about 47% of the total Nr emissions measured in this study, while NO and N2O are approximately 23% and 30% of the total Nr emissions, respectively.
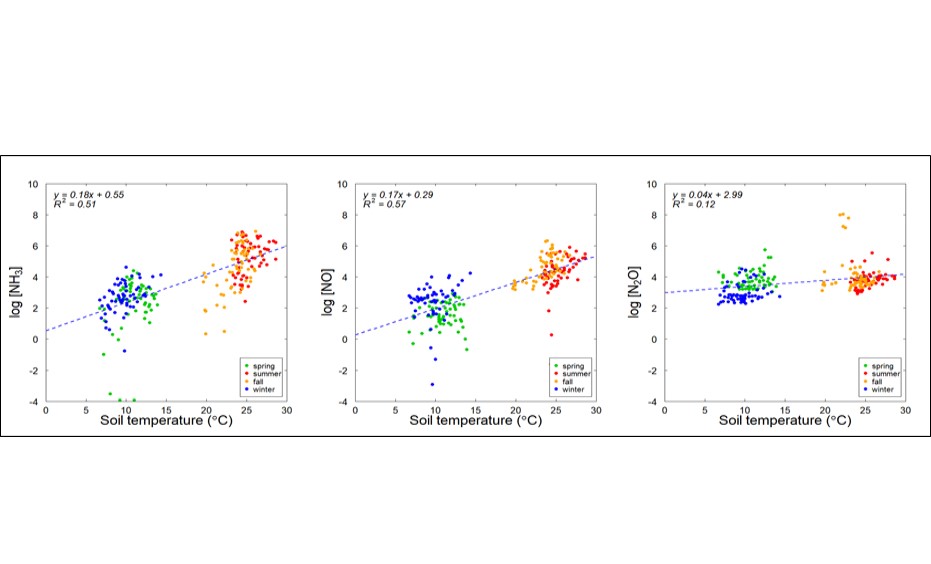
Seasonal emissions of NH3, NO, and N2O from turfgrass system were influenced by several factors. Emission patterns of NH3 and NO were observed to be attributed to the amount of applied N fertilizer (Figure 1) and the soil temperature (Figure 2), as demonstrated by increased emissions in Summer and Spring, and higher emissions following higher amount of applied N-fertilizer. However, the emission pattern of N2O was proved to be less discernible since the relationships between N2O emissions and soil temperature, and between N2O emissions and N-fertilizer are not apparent. The dynamics of soil microbial driven processes is the key for determining the N2O emission profile in a more elaborate manner.
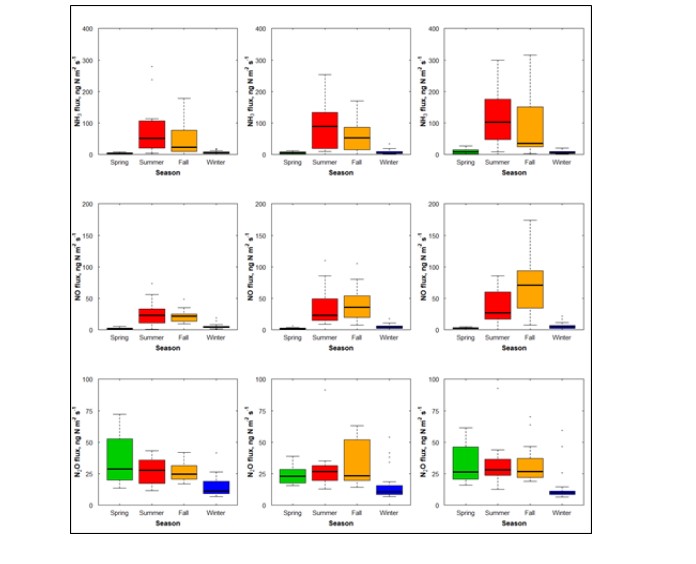
Figure 1. Seasonal variation of NH3 emissions observed from three N-fertilizer treatments: (left-column) treatment A, (middle) treatment B, and (right) treatment C. Emissions are expressed as ng N m-2 s-1.
An important outcome from this study is to estimate emission factors of NH3, NO, and N2O from turfgrass systems, as these systems currently receive little attention with respect to the Nr emission inventory. Collectively, the results suggest that Nr emission factors vary from one season to another, and are dependent upon N input to the soil, soil conditions, and meteorological parameters. These variables need to be taken into considerations when developing an estimate of Nr emissions from turfgrass system.
The U.S. Department of Agriculture (USDA) Environmental Policy Integrated Climate (EPIC) model is being used to simulate plant demand-driven fertilizer applications to commercial turfgrass. This outcome from the model simulations can be coupled with a process-based air quality model to produce continental-scale reactive nitrogen species emission estimates. Regional turfgrass reactive nitrogen compounds emissions are driven by the timing and amount of inorganic N fertilizer applied, soil processes, local meteorology, and ambient air concentrations. This modeling framework will provide a more dynamic, flexible, and spatially and temporally resolved estimate of reactive nitrogen emissions from turf grass and help validate measured reactive nitrogen emission inventories.References
Nahas, A., Walker, J. T., Yelverton, F., and Aneja, V.P. 2020. “Seasonal emissions of Ammonia, Nitric Oxide, and Nitrous Oxide using Dynamic Chamber Measurement Technique on Turfgrass Systems”. In preparation.
Nahas, A., J.T. Walker, F. Yelverton and V. P. Aneja, 2018. “Characterization of Reactive Nitrogen Emissions from Turfgrass”, American Geophysical Union Fall Meeting, Washington, DC, December 10-14, 2018.